Archives
- 2018-07
- 2019-04
- 2019-05
- 2019-06
- 2019-07
- 2019-08
- 2019-09
- 2019-10
- 2019-11
- 2019-12
- 2020-01
- 2020-02
- 2020-03
- 2020-04
- 2020-05
- 2020-06
- 2020-07
- 2020-08
- 2020-09
- 2020-10
- 2020-11
- 2020-12
- 2021-01
- 2021-02
- 2021-03
- 2021-04
- 2021-05
- 2021-06
- 2021-07
- 2021-08
- 2021-09
- 2021-10
- 2021-11
- 2021-12
- 2022-01
- 2022-02
- 2022-03
- 2022-04
- 2022-05
- 2022-06
- 2022-07
- 2022-08
- 2022-09
- 2022-10
- 2022-11
- 2022-12
- 2023-01
- 2023-02
- 2023-03
- 2023-04
- 2023-05
- 2023-06
- 2023-07
- 2023-08
- 2023-09
- 2023-10
- 2023-11
- 2023-12
- 2024-01
- 2024-02
- 2024-03
- 2024-04
-
Glucose permeability studies demonstrated a high
2024-04-19
Glucose permeability studies demonstrated a high glucose flux through the SF films used in the present work. Recent diffusion studies, including small molecular drugs and oxygen permeation through SF membranes [32], further support the quality of SF and its possible application as a material for bio
-
br Experimental section br Results and discussion br Conclus
2024-04-19
Experimental section Results and discussion Conclusion Acknowledgements This work was financially supported by Recruitment Program of Global Experts, and the Director Foundation of XTIPC, CAS, Grant No. 2015RC011. This work was also financially supported by Natural Science Foundation of
-
Brain damage diseases such as cerebral ischemia brain trauma
2024-04-19
Brain damage diseases, such as cerebral ischemia, brain trauma and infection are characterized by the increase in oxygen free radicals. Therefore, deeper understanding of the character and mechanism of free radical damage in the brain is important to ultimately relieve diseases and promote recovery
-
Although CP has been an important research subject in
2024-04-19
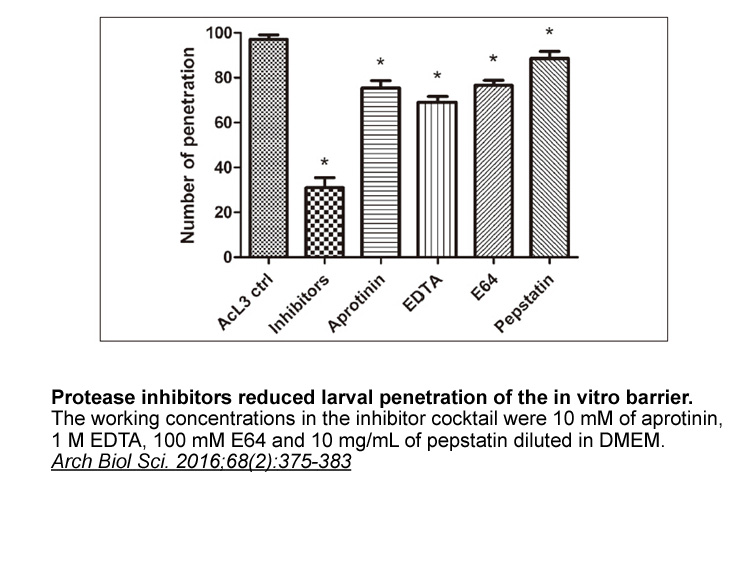
Although CP has been an important research subject in the field of antigen processing and presentation, the mechanisms of presentation of intracellular Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO) by MHC II received considerably less attention. Interestingly, pioneering work by the Münz la
-
Compound was synthesized by reacting amino methoxybenzoate
2024-04-19
Compound 31 was synthesized by reacting 5-amino-2-methoxybenzoate (28) with ethanesulfonyl chloride in the presence of pyridine in DCM to give sulfonamide 29. Hydrolysis of 29 by aqueous sodium hydroxide afforded the Syringin 30 which was converted to the acid chloride by heating in thionyl chloride
-
filipin australia br Experimental EPR spectra of probes a
2024-04-19
Experimental EPR spectra of probes 1a–f were recorded on a Bruker EMX-1572 operating at X-band (9.0–9.9 GHz), at 21 ± 1 °C. The EPR parameters were the same in all experiments: microwave power, 1 mW; modulation amplitude, 5 G; time constant, 10.24 ms; and conversion time, 40.96 ms. Reduced gluta
-
Methods We conducted a secondary analysis of an extant cohor
2024-04-19

Methods We conducted a secondary analysis of an extant cohort of 580 infants hylaxis, premature rupture of membranes, chorioamnionitis, and to prolong pregnancy (latency). Maternal antibiotic exposure did not include GW-1100 synthesis given after the time of delivery or antibiotic initiated by sur
-
AhR was proven to participate in carcinogenesis Its high
2024-04-18
AhR was proven to participate in carcinogenesis. Its high expression was demonstrated in a variety of tumors, i.e. pancreatic, prostate, urinary tract, lung and papillary thyroid carcinoma (Safe et al., 2013, Mian et al., 2014). However, cell line studies showed variable AhR levels/responses. AhR kn
-
HT receptors are distributed throughout the
2024-04-18
5-HT3 receptors are distributed throughout the brain, within the brainstem (e.g., nucleus tractus solitarius, area postrema and spinal trigeminal nucleus) and Zoniporide dihydrochloride receptor (e.g., hippocampus, amygdala, nucleus accumbens, putamen and caudate) (Abi-Dargham et al., 1993, Barnes e
-
The synthetic route to M developed by
2024-04-18

The synthetic route to M100907 developed by Rice was utilized, however, the chiral resolution was carried out at an earlier stage to provide the possibility of introducing different substituents onto the piperidinyl group. The conditions for this resolution were different from previously reported.
-
To better understand the pathophysiology of ASDs we
2024-04-18
To better understand the pathophysiology of ASDs, we would need comprehensive information on a) the functions of ASD-associated proteins in the brain, b) how mutations affect the expression level and function of these proteins, c) how mutations affect their function in neurons, and d) how changed ne
-
br Methods br Results From a study population
2024-04-18
Methods Results From a study population of 7069 patients, a total of 149 falls were reported during the study MHY1485 for an incidence rate (IR) of 5.2 falls per 1000 patient-days (PD), 95% confidence interval (CI) 4.4/1000 PD–6.1/1000 PD. The incidence rate ratio (IRR) for patients in the AC
-
SynaptoRedTM C2 Renal Cell Carcinoma RCC is a lethal cancer
2024-04-18
Renal Cell Carcinoma (RCC) is a lethal cancer with bad prognosis due to development of chemoresistance and recurrence of an aggressive tumor with increased tumor-angiogenesis and metastasis [7]. We have found Axl and Gas6 to be differentially presented in RCC subtypes, and Axl to correlate to tumor
-
An AXL decoy receptor with enhanced GAS binding properties M
2024-04-18
An AXL decoy receptor with enhanced GAS6-binding properties, MYD1, was engineered as a therapeutic tool to disrupt GAS6/AXL signaling in vivo (Kariolis et al., 2014). MYD1 was shown to block metastasis of human ovarian cancer Cryptochlorogenic acid and a murine breast cancer cell line in grafting a
-
br Conflicts of interest br
2024-04-17
Conflicts of interest Financial support This work was supported by grants from the Fondazione Cariplo [Grant number 2011-0463] (Carini); and by Funds for Original Research of the Università del Piemonte Orientale (2016, Project: Carini-Boldorini). The sponsors had no involvement in study desig